Pharmacological enhancement of tissue expansion for breast reconstruction
E. Bertulla, E. Raposio Department of Surgical Sciences and Integrated Diagnostics (DISC), University of Genova, Genoa, Italy. elisa.bertulla@gmail.com
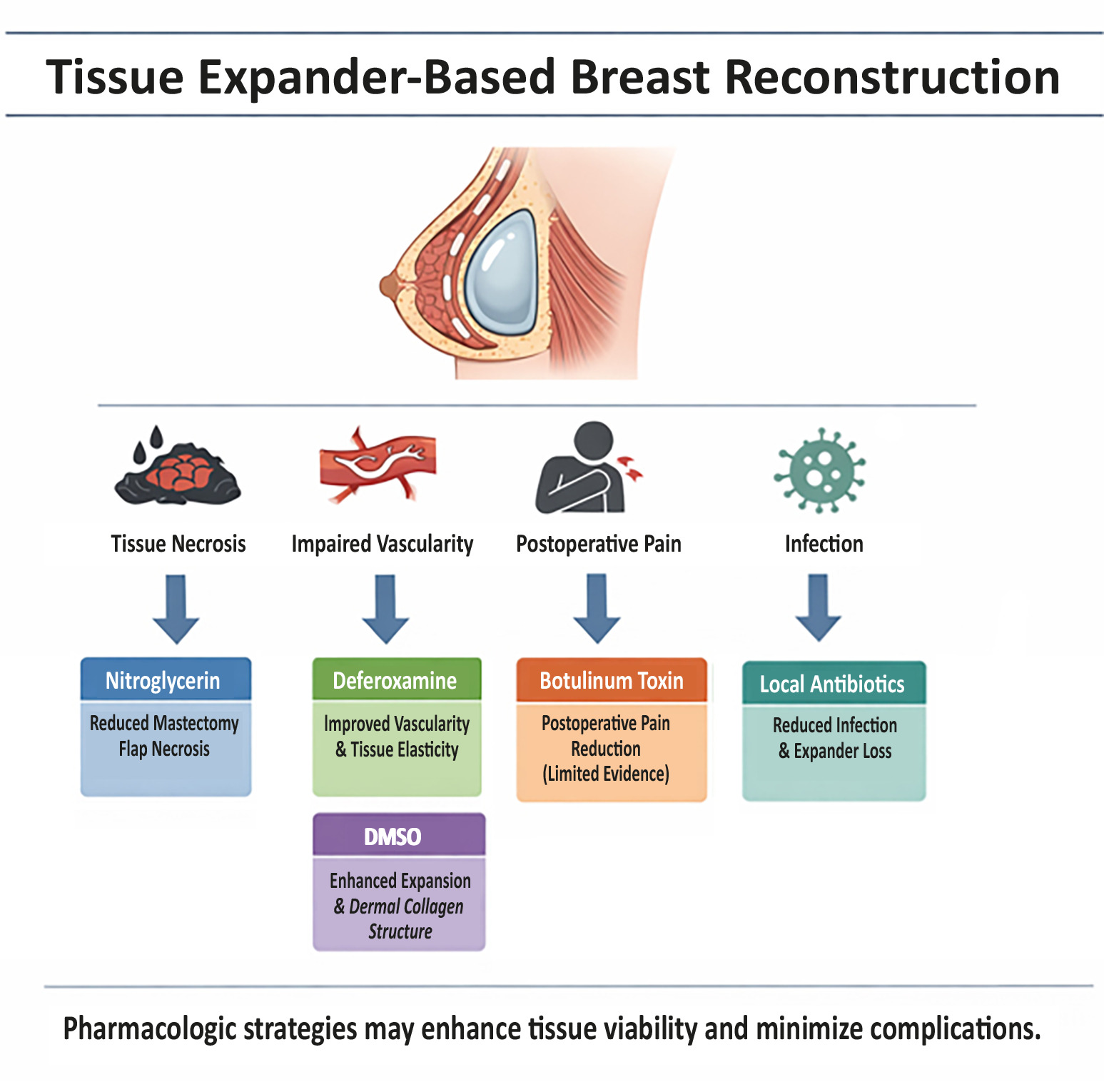
Tissue expander-based breast reconstruction is widely used following mastectomy due to its relative technical simplicity, minimal additional anesthesia, and reversibility. However, complications such as tissue necrosis, impaired vascularity, postoperative pain, and infections are frequent. Pharmacological interventions have been explored to improve tissue viability and reduce these complications. A literature search was performed in PubMed, Scopus, and Cochrane Library, focusing on clinical and preclinical studies investigating pharmacologic agents applied topically or systemically in tissue expander breast reconstruction. Studies on autologous reconstruction, direct-to-implant procedures, cosmetic surgeries, systemic antibiotic prophylaxis, or unrelated drug effects were excluded. Eight studies met the inclusion criteria, including retrospective cohort studies, prospective trials, randomized controlled trials, and preclinical investigations. Agents examined included botulinum toxin, deferoxamine, nitroglycerine, dimethyl sulfoxide, and local antibiotics [absorbable beads or polymethyl methacrylate (PMMA) disks]. Botulinum toxin showed limited evidence for postoperative pain reduction. Deferoxamine increased vascularity and tissue elasticity, especially in irradiated tissue. Nitroglycerine paste reduced mastectomy flap necrosis. Topical Dimethyl sulfoxide (DMSO) improved expansion dynamics and dermal collagen architecture. Local antibiotic delivery significantly decreased infection rates and expander loss, including in patients with skin necrosis.
Overall, pharmacological strategies demonstrate potential to enhance tissue viability, minimize complications, and optimize outcomes in tissue expander-based breast reconstruction. Local antibiotic prophylaxis, topical agents, and vascular modulators appear particularly promising. Nevertheless, evidence remains limited by small sample sizes, heterogeneity of study designs, and the inclusion of some preclinical data. Further prospective, controlled trials are warranted to standardize protocols, confirm safety, and evaluate long-term clinical efficacy.
Graphical Abstract

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License
To cite this article
E. Bertulla, E. Raposio
Pharmacological enhancement of tissue expansion for breast reconstruction
Eur Rev Med Pharmacol Sci
Year: 2026
Vol. 30 - N. 3
Pages: 105-112
DOI: 10.26355/eurrev_202603_37720
Publication History
Submission date: 18 Dec 2025
Revised on: 30 Jan 2026
Accepted on: 04 Feb 2026
Published online: 31 Mar 2026
